Insect Invasion to Guam
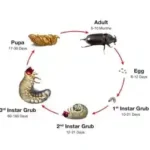







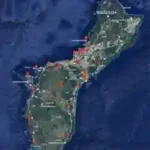
Table of Contents
Share This
“Invasive” organisms are defined by the United States Department of Agriculture, under Executive Order No. 13112, as:
- non-native (or alien) to the ecosystem under consideration.
- introduction causes, or is likely to cause, economic or environmental harm or harm to human health.
Islands are more susceptible to invasive species than larger land masses because island ecosystems evolved in relative geographic isolation. In the distant past, vast mountains and oceans proved sufficient to prevent the migration of even the hardiest of species. Global commerce and travel enable people and cargo to cross these great distances on ships and planes, often taking hitchhikers on the journey.
Guam has become infamous since the accidental introduction of the brown treesnake in the 1940s led to the decimation of the native bird population. Perhaps if the native forest bird population was still thriving on Guam, the insect population would be better controlled.
The following highlights the lesser known insect species that have created some of the worst environmental and economic impacts on Guam.
Coconut Rhinoceros Beetle
The coconut rhinoceros beetle (Oryctes rhinoceros or CRB) is native to Asia, from India to Indonesia. In 2007, the beetles were first identified in the village of Tumon. The destruction of Typhoon Dolphin in 2015 created an unmanageable amount of green waste left to accumulate and decay.This provided suitable breeding grounds for the beetles to reproduce, flourish and establish a growing population. CRB on Guam has been identified to be a unique biotype (renamed CRB-G) resistant to traditional viral biological control agents.
Eradication is now unlikely in 2021 and long-term pest management is needed to prevent CRB from moving to neighboring islands. CRB-G has made its way onto other islands in the region, Hawaii and Rota. The infestation in Hawaii was found in 2013 along a runway at Hickam Air Force Base. More recently in 2017, CRB was discovered in Rota, the southernmost island of the United States Commonwealth of the Northern Mariana Islands. The Department of Agriculture, Biosecurity Division manages CRB pheromone traps around Guam’s major ports of entry, the Port Authority of Guam (commercial seaport) and the AB Won Pat International Airport Authority Guam. Current work has removed CRB breeding sites, over 200 dead standing coconut, other palm trees and green waste piles, around the ports of entry and surrounding loading areas. Ongoing research at the University of Guam in collaboration with international partners work towards a viable biocontrol option.
The CRB goes through a complete life cycle from an egg to larvae to pupa and then to an adult (Figure 1). The larvae (called “grubs”) resemble large, fat, white worms that range up to 10 centimeters long and are found in shallow soil. The larvae body forms a C shape with three pairs of segmented legs on both sides of the body. The adult beetles range from 3.0 to 6.3 centimeters long and are either black or brown, with a prominent horn on their head. Adult male and female beetles can be easily distinguished by identifying their abdomens and the size of their single predominant horn. The tip of the abdomen of an adult female has brown hairs which the males do not possess. Adult male beetle horn size is occasionally longer than that of the adult female. CRB horns are used for leverage and for burrowing into coconut palm crowns.
CRBs feed on the sweet sap in the heart of the palm. After feeding on sap, the beetles search for a suitable breeding ground. After breeding is complete, the female lays her eggs in dead standing coconut trees or in piles of decaying organic matter. The eggs hatch into grubs which feed on the decaying plant matter. After feeding for 60 days, the grubs metamorphoses into a pupa, and develops for up to one month before emerging as an adult beetle.
Flame Tree Looper
The Flame Tree Looper (Pericyma cruegeri) is a moth found in Southeast Asia, Australia and several Pacific Islands in the tropical and subtropical zones. Flame Tree Loopers (Figure 2) were first reported on Guam in 1971. Other reports of the looper have been found in Japan in 1986, on Ishigaki Island and Okinawa Island in 2000. P. cruegeri is a serious pest on the royal poinciana (Delonix regia), yellow flame tree (Peltophorum pterocarpum), and has been found on wild tantan (Desmanthus virgatus), golden shower tree (Cassia fistula), and tangan-tangan (Leucaena leucocephala). On the royal poinciana, they feed on the foliage of the tree and are abundant during the wet season. The caterpillars strip the trees of most of their forage, triggering the creation of smaller leaves, death of small branches, and desynchronized flowering.
Flame tree looper’s yellow or blue-green eggs are laid singly on the flame trees’ leaflets. The eggs hatch within two to three days and caterpillars take at least one month to mature. The head of the caterpillar is distinctively large green with wavy white lines along the sides and top of its body. Once mature, caterpillars pupate on the tree in a cocoon made of webbing and leaves. The moth emerges in 12 days.
The adult moths are dark grey in color, with a wingspan of 3.5 to 4.0 cm. Within the same sex they show minor differences in variation of color. Male and female sex can be determined by examining the antennae and hair growth on the limbs. The male’s antennae are thickened for approximately two-thirds of their length, while the female’s antennae are thin, simple and straight. The hairs on the limbs of males are in a dense profusion and are elongated, while females’ hairs are shorter and not as tightly clustered.
Asian Cycad Scale Insect
Native to Thailand and some parts of China, the armored cycad-specific scale insect (Aulacaspis yasumatsui or CAS) was first detected on Guam in 2003. Since that time, the formerly most abundant tree species, Cycas micronesica, has declined by an estimated 90 percent island-wide (Figure 3). First noticed on ornamental Cycas revoluta plants in Tumon, a failure to quickly isolate the initial infestation led to the armored scale spreading to nearby forests where C. micronesica plants (locally known as fadang) were abundant. Upon finding the vast forests of fadang on Guam, it must have seemed like an all-you-can-eat buffet for the insects. Fadang is the only native host plant for this armored scale insect. Feeding on all parts of the plant and being very small (less than 5 mm) has made controlling this alien herbivore increasingly difficult.
In its native range, this scale insect is a natural pest of various cycad species. Throughout their native range, their populations are kept in check by various predatory insects. On Guam however, these predators are not present.
Biological control efforts to control CAS have been met with mixed results. Two parasitoid wasp species, Coccobius fulvus and Aphytis lignanensis, failed to establish on Guam when introduced in 2005 (Figure 4). The predatory lady beetle, Rhyzobius lophanthae, was also introduced in 2005 and readily established throughout Guam (Figure 5). The lady beetle has helped reduce mortality from CAS herbivory but has not been effective in protecting seedlings and juvenile plants.
As a result, the once iconic fadang has become a threatened species. Until the ongoing herbivory by CAS is under control, reintroductions of C. micronesica will be futile. Off island collections of C. micronesica exist which can be utilized for future reintroductions.
Cycad Blue Butterfly
Another alien herbivore has also added to the ongoing assault of the cycad trees. The Cycad blue butterfly (Chilades pandava) is native to India, Sri Lanka, Myanmar, United Arab Emirates, Indochina, Peninsular Malaysia, Singapore, Taiwan, Java, Sumatra and the Philippines. This butterfly was first detected on Guam in 2005. This lycaenid butterfly is a specialist herbivore of Cycas plants. Although still harmful to C. micronesica, herbivory from this pest is not fatal. As with CAS, C. micronesica is the only native host on Guam for this butterfly, making it a significant pest even if it is not fatal.
On the upper side of the wings, the male is blue with thin black borders on both wings and a black spot on the corner of the hindwing (a “tornal” spot). The female is a paler blue with broad borders on the forewing, a series of submarginal spots on the hindwing with the second spot crowned in orange. The female oviposits on young emerging growth of various Cycas species. After two days, the eggs emerge as caterpillars and consume leaf tissue on the soft and emerging leaflets (Figure 6) As a result, the plant is left only with the rachis.
In addition, several ant species existed on Guam prior to the butterfly’s invasion. These ants work together with caterpillars by protecting the caterpillars from would be predators. In return for protection, the ants receive a nectar reward from the caterpillars, thus adding to the assaults on Guam’s native cycad. Biological control of the Cycad Blue butterfly has not been attempted to date due to concerns over native butterfly species.
Psyllids
One of Guam’s smaller invasive species is the Asian Citrus Psyllid (Diaphorina citri) , which is native to Asia but prevalent in the citrus growing areas of the Continental United States. These citrus plant pests are most noted for carrying the bacterium Candidatus Liberibacter spp. which is the causal agent of Citrus Greening disease. Citrus Greening disease causes Guam’s citrus trees to lose color, form misshapen fruit, and to die in some cases. Since their introduction, the psyllid population has posed a threat to Guam’s citrus trees.
The Asian Citrus Psyllid was first found on Guam in 2008 by RK Campbell. The psyllids may have been introduced by infested plant material, which is a common trend amongst invasive insects. Psyllids prefer temperatures around 80 Farenheit, which makes Guam’s climate ideal for the insect to thrive and spread the disease.
Two common ways to eradicate agricultural pests are the use of pesticides and biological control agents. Pesticides are more commonly used on citrus farms because of their ease of application over numerous trees. Although they are easy to use, pesticides are costly and may be dangerous. A more cost-effective way to manage Guam’s psyllid population is using biocontrol insects. There are five beetles, brown-lacewing larvae, and other insects that eat psyllids at different stages of their lives. To prevent the spread of this invasive insect, Guam needs to implement a combination of control methods.
Greater Banded Hornet
A relatively new invasive insect is the Greater Banded Hornet (Vespa tropica). It is commonly found in Southeast Asia. The hornet was found on Guam on 12 July 2016 by Christopher Rosario, a University of Guam student. The Greater Banded Hornet (Figure 8) is characterized by a single large band, that wraps around the abdomen of the hornet. It is not every “big bee” that can be seen flying around your home.
Since its introduction to Guam, hornet attacks have been a public problem. In August of 2016, The Guam Daily Post wrote an article about firefighters being attacked by hornets. In this article, they describe how the firefighters were attacked and hospitalized during a dog rescue.
This kind of attack is not uncommon, as hornets are known to attack when their nest is disturbed. The Greater Banded Hornet also attacks bees and wasps. The hornets have been observed catching honey bee foragers, by Guam’s beekeepers.
Because Guam is small and lacks natural predators of the hornet, the hornets have been able to establish their population on Guam. The Greater Banded Hornet can travel miles away from their hive to forage. This ability to travel great distances has resulted in multiple sightings of the hornets (refer to Figure 9). Since their establishment, the hornet has been sighted in the northern part of Guam. If you have seen this invasive species, please call the Guam Department of Agriculture-Biosecurity Division at 671-475-PEST (7378).
Little Fire Ants
Little fire ant, (Wasmannia auropunctata Rodger) (LFA), is an ant species native to Central and South America but has now spread to parts of Africa, North America, Puerto Rico, Israel, Cuba, and several Pacific Islands including Guam. LFAs (Figure 10) were first detected in a karst limestone forest adjacent to a green waste landfill on Guam in November of 2011. This aggressive ant has since spread to all parts of the island
The devastating effects that LFA has had on agriculture and local forest ecosystems outside of its native range are also likely to occur on Guam. Unfortunately, LFA has now spread all over Guam and infestations are particularly heavy in areas close to streams.
The sting of LFA is very painful and those who may be allergic to stings are at particular risk. The sting of LFA may also cause blindness in indigenous and domesticated animals.
Recent efforts to eradicate LFA with techniques utilizing toxicants in gel-based matrices developed by the Hawaii Ant Lab (littlefireants.com) has proven to be effective at controlling the LFA in small controlled areas. Although manageable, the movement of infested green waste and live plants fuel the spread of LFA around Guam. LFA has now made its way to the island of Yap via infested cargo from Guam. LFA can be reported to the Guam Department of Agriculture, Biosecurity Division at 475-PEST(7378) and more information can be found here.
Lobate lac scale
Lobate lac scale (Paratachardina pseudolobata) is a pest of woody trees with a wide host range including hibiscus, ficus, acacia, mango, avocado, guava and some orchids. The pest has been identified in parts of Asia, Australia, Florida and more recently in Hawaii in 2012. The female scale produces lac, a waxy resin structure which resembles a tiny red-black X-shaped mushroom on the plant surface. Immature insects can produce thread-like structure that enable it to be carried and spread by the wind.
The scale was discovered by Aubrey Moore in 2018 at the University of Guam’s Yigo Experiment Station. Initially identified on hibiscus and joga trees at the Experiment Station, community reports and surveys by Guam Department of Agriculture, Biosecurity Division have found the scale throughout the island. The scale can be found young acacia trees and new tree growth in the Cotal Conservation Area on Cross Island Road in Sånta Rita-Sumai. The scales feed off nutrients from plant phloem and heavy infestations lead to the growth of sooty mold (which grows off the insect’s excrement), loss of leaves, stem dieback and death of young trees. Although the impact of the lobate lac scale is undetermined on Guam, Hawai’i has reported significant losses in banyan (ficus) trees.
About the authors

Benjamin Deloso was a graduate student majoring in Environmental Science at the University of Guam. He holds a BA in Biology with a minor in Psychology from the University of North Carolina Wilmington. During his time in college he became interested in botany and horticulture, and became enamored with a particular group of plants called cycads. Originally from Guam, his horticulture experience and botanical exploits led him to return to Guam to study under cycad expert Dr. Thomas Marler. Since arriving at UOG in 2016 he has designed and maintained a cycad teaching garden located near the Agriculture and Life Science building as well as conducted fieldwork in cycad habitats in Yap, Palau, Rota, and the Philippines. Grateful for the opportunities to study cycads in the tropics, Benjamin hopes to become a world authority on cycads in the future. He wrote the sections on cycad-specific insect pests.
Chieriel Desamito was a graduate student majoring in Sustainable Agriculture, Food and Natural Resource (SAFNR) at the University of Guam. He holds a Tropical Agriculture Applied degree from the University of Guam in 2016. During his time in college, he took an interest in Soil Science after taking classes regarding soil health and fertility. He was currently working as a Research Associate for the UOG soil laboratory and as a teaching assistant for Dr. Golabi’s lab on the Fundamentals of Soil Science. He was conducting research with Dr. Golabi on the effects of biochar, compost, and fertilizer application as a soil amendment, in order to promote agricultural sustainability and reduce greenhouse gas emissions from intensive tillage practices. He was pursuing research opportunities with Dr. Golabi to expand his knowledge on Guam’s soils. He wrote the sections on CRB and Flame Tree Loopers.


Kaleb Leon Guerrero graduated with a Research in Tropical Agriculture degree from the University of Guam in 2017. He was working towards his masters degree in Sustainable Agriculture, Family Science, and Nutrition at the University of Guam. He was working at the University of Guam as a Research Associate for the entomology lab, and an adjunct professor for the BI-100L class. During his time internship at UOG in 2008, he became interested in the field of entomology. Since his senior year in college, he has conducted research in entomology under Dr. Ross Miller. He has conducted field work on Guam, Saipan, Tinian, and Rota. Grateful for the opportunities his work in entomology has presented, he is furthering his knowledge on different species of insects and conducting more field work. He wrote the sections on Psyllids and the Greater Banded Hornet.
Editor’s note: These authors were beginning graduate students in 2018 taking a course in scientific writing at the University of Guam. This article was assigned to provide the student with practice in communicating science to non-scientists. The student chose the topic which is related either to their thesis project or work experience. The instructor in the course is Dr. Laurie Raymundo, a UOG Marine Laboratory faculty member. The information in this entry was updated in 2021 by Dr. G. Curt Fiedler, Professor of Biology and Editor of Micronesica at College of Natural and Applied Sciences University of Guam.
For further reading
Aubert, B. “Trioza erytreae Del Guercio and Diaphorina citri Kuwayama (Homoptera Psylloidea), the Two Vectors of Citrus Greening Disease: Biological Aspects and Possible Control Strategies.” Fruits 42, no. 3 (1987): 149-162.
Bové, JM. “Huanglongbing: A Destructive, Newly-emerging, Century-old Disease of Citrus.” Journal of Plant Pathology 88, no. 1 (2006): 7-37.
Campbell, RK. “Incursion of Citrus Psyllid in Guam.” Pest Alert, no. 40 (2008): 1.
Doane, RW. “How Oryctes Rhinoceros, A Dynastid Beetle, Uses Its Horn.” Science 38, no. 990 (1913): 883.
Dornberg, Mike. “Coconut Rhinoceros Beetle – Oryctes rhinoceros.” Featured Creatures, University of Florida, July 2015.
Giblin-Davis, Robin M. “Borers of Palms.” In Insects on Palms, by Forrest W. Howard, Dave Moore, Robin M. Giblin-Davis, and Reynaldo G. Abad, 267-304. Wallingford: CABI Publishing, 2001.
Gottwald, Tim R., John V. da Graça, and Renato B. Bassanezi. “Citrus Huanglongbing: The Pathogen and Its Impact.” Plant Health Progress 8, no. 1 (2007).
Halbert, Susan E., and Keremane L. Manjunath. “Asian Citrus Psyllids (Sternorrhyncha: Psyllidae) and Greening Disease of Citrus: A Literature Review and Assessment of Risk in Florida.” Florida Entomologist 87, no. 3 (2004): 330-353.
Halbert, Susan E., Keremane L. Manjunath, Chandrika Ramadugu, Matthew W. Brodie, Susan E. Webb, and Richard F. Lee. “Trailers Transporting Oranges to Processing Plants Move Asian Citrus Psyllids.” Florida Entomologist 93, no. 1 (2010): 33-38.
Liu, Ying H., and James H Tsai. “Effects of Temperature on Biology and Life Table Parameters of the Asian Citrus Psyllid, Diaphorina citri Kuwayama (Homoptera: Psyllidae).” Annals of Applied Biology 137, no. 3 (2000): 201-206.
Marler, Thomas E., and John H. Lawrence. “Demography of Cycas micronesica on Guam Following Introduction of the Armored Scale Aulacaspis yasumatsui.” Journal of Tropical Ecology 23, no. 3 (2012): 233-242.
Marler, Thomas E., and R. Muniappan. “Pests of Cycas micronesica Leaf, Stem, and Male Reproductive Tissues with Notes on Current Threat Status.” Micronesica 39, no. 1 (2006): 1-9.
Marler, Thomas E., Ross Miller, and Aubrey Moore. “Vertical Stratification of Predation on Aulacaspis yasumatsui Infesting Cycas micronesica Seedlings.” HortScience 48, no. 1 (2013): 60-62.
McCaffrey, Sarah, and Ken Walker. “Vespa tropica.” Australian Biosecurity, Pest and Disease Image Library (PaDIL), 8 March 2012.
Michaud, JP. “Biological Control of Asian Citrus Psyllid, Diaphorina Citri (Hemiptera: Psyllidae) in Florida: A Preliminary Report.” Entomological News 113, no. 3 (2002): 216-222.
Mishra, RC, and Rajesh Garg, eds. Perspectives in Indian Apiculture. Bikaner: Agro Botanica, 1998.
Rosario, Christopher A., Lee Roy Sablan, Aubrey Moore, and Ross H. Miller. Greater Banded Hornet, Vespa tropica (Hymenoptera: Vespidae). Guam New Invasive Species Alert No. 2016-01. Mangilao: College of Natural & Applied Sciences, University of Guam, 2016.
